High performance and high HbA1c: fitness does not guarantee protection from poor sleep
Here’s a conundrum.
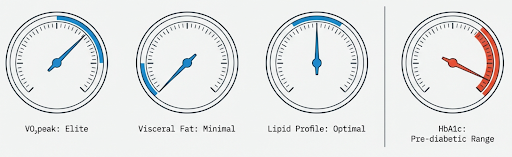
You are fit, with high VO2peak, low visceral fat, and reassuring lipids.
Yet HbA1c sits high normal, or drifts into the prediabetic range.
The default explanation is insulin resistance.
In some lean, high performing people, that explanation does not tally with the rest of the data.
I think this is one of the more interesting mismatches I see in my practice. You can have obvious signs of good “peripheral” metabolic health and still see suboptimal blood sugar levels.. The current evidence suggests that sleep, stress, and insulin secretion may explain part of that gap, although the phenotype is not yet defined as a formal syndrome in the literature [1].
A brief primer
Glucose is your main circulating fuel. It rises after meals, especially after carbohydrate, and falls as tissues take it up or store it.
Insulin is the hormone that keeps that rise controlled. It helps muscle and other tissues take up glucose, and it suppresses glucose output from the liver.
HbA1c is not a real time marker. It reflects average glucose exposure over roughly the last 2 to 3 months, because glucose slowly binds to haemoglobin over the lifespan of red blood cells.
The foundational physiology still matters
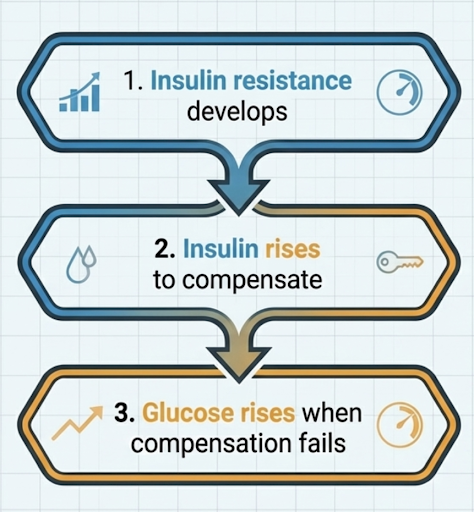
Insulin resistance develops.
Insulin rises to compensate.
Glucose rises when compensation fails.
That model explains a large chunk of the path to type 2 diabetes. It is particularly relevant to sedentary populations, obesity, and the classic hyperinsulinaemic pattern. But it does not explain why some active, lean, high-performing people show a mildly elevated HbA1c despite otherwise reassuring markers. That is where there is some nuance.
A better place to start is insulin secretion
In an interesting study, Kamba et al. undertook a cross sectional look at 1,071 Japanese adults not taking diabetes medication or steroids [1]. They measured cortisol and calculated fasting surrogates for insulin resistance and insulin secretion using HOMA indices. Higher cortisol was associated with lower HOMA β, but not with HOMA IR.
That does mean to say that elevated cortisol is damaging beta cells.
It also does not explain what happens during a meal, during exercise, or across the full day. It’s important to note that HOMA measures are fasting surrogates, not dynamic functional blood tests. But the paper does support a narrower and clinically relevant point that higher cortisol was associated with lower estimated insulin secretion, independent of insulin resistance, in this population.
That is noteworthy because it brings a different perspective.
If insulin secretion is slightly blunted, post meal glucose can run higher without fasting insulin looking abnormal. Over time, these small elevations in glucose may accumulate, even when fasting markers look deceptively normal. This is an inference from the study plus the physiology, not a direct finding from that paper.
Stress is not just a feeling here
The next question is whether stress actually results in long term elevated glucose.
A recent real world study helps unpick this. In 2025, Schrems et al. used ambulatory stress assessment over 3 days and continuous glucose monitoring over 7 days in 116 participants, split into insulin-resistant and insulin-sensitive groups [2]. Daily life stress was associated with higher glucose levels in the insulin-resistant group, but not in the insulin-sensitive controls.
This paper does not show the same effect in highly fit, insulin-sensitive, high VO₂max people. So we can’t overextrapolate here. But it does show that psychological stress can have measurable glycaemic consequences in real life, not just under artificial laboratory conditions. That makes the broader argument more credible.
Sleep is one of the strongest levers
If I had to pick one upstream variable most likely to matter in this scenario, it would be sleep.
A 2022 systematic review and meta-analysis (a review of all papers on a topic) found that sleep restriction reduced insulin sensitivity across multiple assessment methods, including more dynamic blood tests, such as the oral and intravenous glucose tolerance tests, as well as HOMA-IR [3].
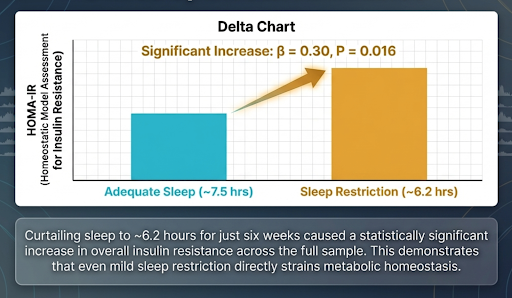
More recent randomised data from 2024 suggest that even mild chronic sleep restriction can impair insulin sensitivity: in a 6-week crossover trial, reducing sleep by about 1.3 hours per night increased fasting insulin and HOMA-IR in women, independent of fat mass changes [4].
These changes are large enough (~20%) to move someone from apparently robust metabolic control into a more fragile state, at least transiently.
Sleep deficits & cortisol
One plausible link between short sleep and impaired glucose regulation is cortisol. Sleep restriction does not simply affect sleep duration. It can also alter neuroendocrine timing, including the daily rhythm of cortisol secretion.
Sleep restriction is not just about “more cortisol”. It is also about when cortisol is elevated. Experimental work shows that restricted sleep can raise late afternoon and evening cortisol, which means the normal diurnal pattern gets distorted rather than simply amplified [5], [6].
Why does that matter?
Because evening and overnight physiology should be gearing us up towards rest and recovery, not toward continued readiness. A later cortisol signal can plausibly contribute to higher glucose production, less stable overnight glucose, and a less favourable fasting picture by morning.
Sleep extension helps
This is the piece that makes this more than just a mechanism.
A 2 week sleep extension trial in chronically sleep deprived (mean sleep duration ~5h 15min) healthy volunteers found that the overall intervention did not improve glucose metabolism in everyone, but only in those who bjectively extended sleep to more than 6 hours per night [7]. Specifically, HOMA-IR fell, early insulin secretion rose, and disposition index, a marker of beta cell function adjusted for insulin sensitivity, improved.
So how does this fit together with foundational physiology?
I would look at it like this. You can have good peripheral (muscle) metabolic capacity and poor central regulation at the same time.
That means:
Muscle insulin sensitivity may be relatively preserved.
Cardiorespiratory fitness may be high.
Sleep may still be short or fragmented.
Stress physiology may still be dysregulated - elevated and/or mistimed cortisol profile
Compensatory insulin secretion may be blunted.
If that happens, HbA1c can drift upward without the classic picture of obvious insulin resistance. That exact phenotype is not formally defined in a single paper. It is a synthesis across clinical experience, combined with the cortisol, stress study, sleep restriction and sleep extension literature above.
What this changes in practice
If you are lean, active, and clearly high performing, but HbA1c does not fit, do not jump straight to “hidden insulin resistance”. Start with four questions:
Is HbA1c reliable here, given iron status, red cell turnover, or haemoglobin traits?
Is the problem fasting glucose, post meal excursions, overnight instability, dawn rise, or a mix?
Is sleep short, fragmented, or irregular, and does glucose track with under recovery?
Does dynamic testing suggest a secretion lag rather than a fasting resistance pattern?
What is your psychological stress and allostatic load?
Is training load and recovery balanced?
Obviously, sleep is not the only factor to consider. Common genetic variations can influence HbA1c levels, through non-glucose pathways, and haemoglobin variants. These can make HbA1c read higher or lower than true glucose exposure would suggest [8], [9].
Genetics may also shape susceptibility. Variation in circadian and beta-cell pathways (such as MTNR1B) has been associated with higher fasting glucose and impaired insulin secretion, so sleep and circadian disruption may be exaggerated by a pre-existing physiological vulnerability in some individuals [10].
This doesn’t mean insulin resistance doesn’t matter. It just may not be the whole story in this subgroup of individuals.
The right tools are usually dynamic
If your goal is to understand the pattern rather than guess at it, use tools that can show physiology in motion.
CGM can show whether the problem is postprandial excursion, overnight instability, or an early morning rise.
Dynamic testing such as an OGTT with insulin or C-peptide can tell you more about early insulin response than fasting insulin alone.
Sleep and recovery data can help you test whether the glycaemic pattern tracks with under-recovery.
Treat HbA1c as a summary marker, not as the full story.
That mindset is especially useful in people who look metabolically well on the surface. The studies above support the physiology behind this approach, although they do not test this exact clinical workflow directly.
What the current evidence does not prove
The evidence does not prove that cortisol alone causes beta cell dysfunction. It does not prove that fixing sleep will normalise HbA1c in every situation. And it does not justify treating every mildly high HbA1c in an athletic person as a sleep problem.
What it does support is a more careful clinical consideration.
Sleep restriction impairs glucose metabolism.
Real world stress can raise glucose in at risk individuals.
Higher physiological cortisol is associated with lower fasting estimated insulin secretion.
And sleep extension can improve insulin sensitivity and early insulin response in some short sleepers.
Bottom line
When an otherwise high-performing individual has an HbA1c that does not fit the rest of the picture, I would not assume insulin resistance is the only explanation.
A more precise interpretation is that glucose regulation may be getting pushed off course by sleep loss, stress physiology, altered cortisol timing, and reduced compensatory insulin secretion. In some people, that may coexist with very good fitness and otherwise reassuring markers. The model is plausible and partly supported, but it is still a synthesis rather than a fully defined clinical entity.
Q&As
How can someone be fit and still have a raised HbA1c?
Because fitness does not guarantee perfect glucose regulation. You can have high VO₂peak, low visceral fat, and good lipids, yet still show a mildly elevated HbA1c if sleep, stress physiology, cortisol timing, or insulin secretion are off.
Does a high normal or mildly raised HbA1c always mean insulin resistance?
No. Insulin resistance is one explanation, but not the only one. In lean, active, high-performing people, the picture can be more nuanced than the classic pathway seen in obesity and sedentary populations.
What does HbA1c actually measure?
HbA1c reflects average glucose exposure over roughly the previous 2 to 3 months. It is not a real-time glucose reading. It is a summary marker, not a full physiological explanation.
What is the usual model for worsening glucose control?
The standard model is that insulin resistance develops first, insulin rises to compensate, and glucose rises when that compensation starts to fail. That still explains a large part of the path to type 2 diabetes. It just does not explain every lean, athletic phenotype.
Could the issue be insulin secretion rather than insulin resistance?
Yes. That is one of the main points of the piece. If insulin secretion is slightly blunted, especially early after meals, glucose can run higher without fasting insulin looking obviously abnormal.
Why does sleep matter so much here?
Because sleep restriction can impair insulin sensitivity even without changes in body fat. It can also alter cortisol timing and make glucose regulation more fragile. In practice, sleep is often one of the most important upstream levers.
Is cortisol the whole explanation?
No. The evidence does not prove that cortisol alone causes beta-cell dysfunction or explains every case. The more balanced view is that altered cortisol timing may be one part of a wider picture involving sleep, stress load, and compensatory insulin secretion.
Can good muscle health and poor glucose regulation coexist?
Yes. That is a useful way to frame this mismatch. Muscle insulin sensitivity and fitness may look strong, while sleep, recovery, stress biology, or insulin secretion are still pulling glucose control in the wrong direction.
Could HbA1c be misleading rather than truly abnormal?
Yes. HbA1c can be distorted by iron status, red cell turnover, haemoglobin variants, and some genetic factors. That means a higher reading does not always equal higher true glucose exposure.
What tests are more useful than guessing?
Dynamic tools are usually more helpful. CGM can show whether the problem is post-meal, overnight, or early morning. An oral glucose tolerance test with insulin or C-peptide can assess insulin response better than fasting insulin alone. Sleep and recovery data can also help link the pattern to under-recovery.
What is the practical takeaway?
Do not assume hidden insulin resistance is the only answer. In a lean, active, high-performing person with an HbA1c that does not fit, it is worth looking at sleep, stress, recovery, insulin secretion, and whether HbA1c is even the right marker to rely on in isolation.
Further Reading
[1] A. Kamba et al., ‘Association between Higher Serum Cortisol Levels and Decreased Insulin Secretion in a General Population’, PLoS ONE, vol. 11, no. 11, p. e0166077, Nov. 2016, doi: 10.1371/journal.pone.0166077.
[2] E. Schrems et al., ‘Daily life stress is linked to increased glucose levels in individuals with insulin resistance: a real-world assessment’, Diabetologia, vol. 68, no. 12, pp. 2709–2718, Dec. 2025, doi: 10.1007/s00125-025-06552-x.
[3] N. Sondrup et al., ‘Effects of sleep manipulation on markers of insulin sensitivity: A systematic review and meta-analysis of randomized controlled trials’, Sleep Med. Rev., vol. 62, p. 101594, Apr. 2022, doi: 10.1016/j.smrv.2022.101594.
[4] F. M. Zuraikat et al., ‘Chronic Insufficient Sleep in Women Impairs Insulin Sensitivity Independent of Adiposity Changes: Results of a Randomized Trial’, Diabetes Care, vol. 47, no. 1, pp. 117–125, Jan. 2024, doi: 10.2337/dc23-1156.
[5] R. Killick, S. Banks, and P. Y. Liu, ‘Implications of Sleep Restriction and Recovery on Metabolic Outcomes’, J. Clin. Endocrinol. Metab., vol. 97, no. 11, pp. 3876–3890, Nov. 2012, doi: 10.1210/jc.2012-1845.
[6] E. C. Hanlon et al., ‘Sleep Restriction Enhances the Daily Rhythm of Circulating Levels of Endocannabinoid 2-Arachidonoylglycerol’, Sleep, vol. 39, no. 3, pp. 653–664, Mar. 2016, doi: 10.5665/sleep.5546.
[7] A. So-ngern, N. Chirakalwasan, S. Saetung, S. Chanprasertyothin, A. Thakkinstian, and S. Reutrakul, ‘Effects of Two-Week Sleep Extension on Glucose Metabolism in Chronically Sleep-Deprived Individuals’, J. Clin. Sleep Med. JCSM Off. Publ. Am. Acad. Sleep Med., vol. 15, no. 5, pp. 711–718, May 2019, doi: 10.5664/jcsm.7758.
[8] E. Wheeler et al., ‘Impact of common genetic determinants of Hemoglobin A1c on type 2 diabetes risk and diagnosis in ancestrally diverse populations: A transethnic genome-wide meta-analysis’, PLOS Med., vol. 14, no. 9, p. e1002383, Sep. 2017, doi: 10.1371/journal.pmed.1002383.
[9] R. R. Little and W. L. Roberts, ‘A review of variant hemoglobins interfering with hemoglobin A1c measurement’, J. Diabetes Sci. Technol., vol. 3, no. 3, pp. 446–451, May 2009, doi: 10.1177/193229680900300307.
[10] J. M. Lane et al., ‘Impact of Common Diabetes Risk Variant in MTNR1B on Sleep, Circadian, and Melatonin Physiology’, Diabetes, vol. 65, no. 6, pp. 1741–1751, Jun. 2016, doi: 10.2337/db15-0999.