NAD⁺ in humans: can you raise it and does it matter clinically
NAD⁺ biology is strong.
That is exactly why the marketing gets ahead of the evidence.
Human trials show you can raise a blood marker.
They do not yet show reliable functional benefit in healthy older adults.
IV NAD⁺ sits lowest on the evidence ladder, despite the loudest claims.
The science behind NAD⁺ and ageing is genuinely interesting. That is part of the problem.
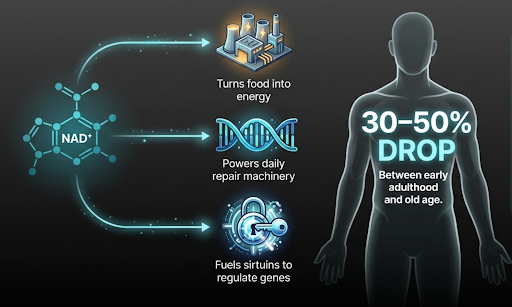
NAD⁺ has become one of the most talked about ideas in longevity. NAD⁺ is not a single-purpose molecule. At the most basic level, it helps cells turn food into usable energy.
But it also does more than that.
It is linked to aging in several critical ways:
to repair damage that accumulates in your DNA every day
to power a family of proteins (sirtuins) that regulate how genes are read
mitochondrial dysfunction
chronic inflammation
The theory goes that NAD⁺ falls with age, supplements raise it, and raising it should improve energy, metabolism, brain function, and slow down the ageing process.
When the biology seems compelling, the commercial language can often move quickly and extrapolate from concept to clinical certainty rapidly.
IV NAD⁺ drip clinics have proliferated, marketing anti-aging, cellular rejuvenation and cognitive restoration. Oral supplements, such as NR, NMN, or "mixed-pathway supplements" sit prominently in longevity conversations.
The biological rationale gets invoked as though it were proven clinical evidence, and the gap between the two gets understated.
The question most people are asking, though, is “does it work?”, is the wrong starting point for where the evidence is at.
It needs decomposing. There are three separate questions here, and right now they have three different answers.
Why NAD⁺ Falls with Age: The Biology in Brief
Because NAD⁺ gets used up in multiple processes in cells every day, the body has to keep making it continuously.
Most of this production works like an internal recycling system, the “salvage pathway”: as it gets used, NAD⁺ is broken down into a simpler form, then reformed back into NAD⁺ [1].
Three significant factors reduce as we age:
The enzyme (NAMPT) that controls the rate of this recycling becomes less active as we age, which means the recycling rate slows down and NAD⁺ levels drop [2].
At the same time, a separate enzyme (CD38) that uses up NAD⁺, becomes more active with age. This is driven by the low-grade inflammation that builds up in older tissues [3].
Because DNA damage accumulates over decades as awe age, the repair machinery that consumes NAD⁺ is increasingly in demand over the years [4].
The result is an estimated 30–50% fall in NAD⁺ levels between early adulthood and old age [4].
That is the biological case for doing something. It is strong, but it doesn’t answer the practical question of whether taking NAD⁺ or one of its building blocks (precursors) as a supplement actually reverse this depletion that we see with advancing age. Supplying the raw material (NAD⁺) is one problem. Fixing the machinery that increasingly consumes it with age, is a different one.
A Brief Taxonomy of the Supplements
The precursors differ in where they enter the NAD⁺ pathway and in how much clinical evidence accompanies them.
Niacin, also called nicotinic acid, and simply a form of vitamin B3, has been used clinically for decades and is highly effective at raising NAD⁺ levels. It is less used currently as it causes an unpleasant skin flushing reaction in most people who take it, and more importantly, the large body of clinical trials testing niacin for heart disease (the main clinical indication it was used for) has been disappointing and has not reduced cardiovascular events in the way earlier trials once suggested it might [5].
Nicotinamide (NAM) is a related compound that can also feed the NAD⁺ recycling system. One issue, however, is that at higher doses it appears to block the very enzymes involved in gene regulation and DNA repair that increasing NAD⁺ is supposed to support [6].
These are the older supplement approaches. A dedicated randomised controlled trial gave older adults with impaired physical function a combination of the three classical NAD⁺ building blocks: the amino acid tryptophan, niacin, and nicotinamide. The researchers then measured the effect on NAD⁺ levels in muscle, on how well their cells' energy-generating machinery worked, and on their physical performance. There was no improvement on any of those measures [7].
That is a disappointing null result, tested in exactly the population these supplements are most widely targeted to support.
Nicotinamide riboside (NR) and nicotinamide mononucleotide (NMN) are the two supplements most widely sold as NAD⁺ boosters. Both are forms of vitamin B3 that the body can convert into NAD⁺ through its internal recycling system.
NR is the most extensively studied of the newer NAD⁺ precursors in human clinical settings.
Multiple trials consistently demonstrate that oral NR at doses of 500-2,000 mg/day elevates whole-blood NAD⁺ substantially above baseline within 2-4 weeks, with dose-dependent increases of approximately 40-150% [8], [9].
NMN is positioned one step closer to NAD⁺ in the biosynthetic pathway than NR. Despite widespread advocacy, partly driven by high-profile scientist endorsements, the clinical evidence base is modest, and head-to-head comparisons with NR have not definitively established any advantage [10].
Again, multiple studies confirm that oral NMN at 250-1,000 mg/day elevates blood NAD⁺ by approximately 100–140% [11], [12]
A recent study tracking how these compounds actually behave once swallowed adds a complicating twist: a significant proportion of both NR and NMN appears to be converted by gut bacteria into plain niacin, the old, cheap form of vitamin B3, before it even reaches the bloodstream [10].
In other words, part of what is being sold as a sophisticated next-generation approach may, in practice, be working through the same route as a supplement you could buy for pennies.
Multi-component formulations are a distinct approach worth noting which sit outside the single-precursor model.
The theory is built on a systems biology rationale: meaning instead of simply supplying the body with more NAD⁺ raw material and hoping the cell does the rest, it attempts to fix several of the regulatory mechanisms that break down with age simultaneously.
One formulation combines NAM as the NAD⁺ building block with additional active ingredients to target different points of NAD⁺ decline: plant compounds to block CD38 (the enzyme that degrades NAD⁺); flavonoids and other compounds to upregulate NAMPT (the rate-limiting recycling enzyme); and compounds found in green tea, to inhibit the enzyme responsible for chemically tagging NAM for excretion rather than recycling it back into NAD⁺.
In other words, the formulation tries to supply the substrate, slow the degradation, accelerate the recycling, and block the disposal route, all at once.
The logic is coherent and addresses a genuine limitation of single-precursor strategies.
One published double-blind, randomised crossover trial, in which neither participants nor researchers knew which treatment was which, and each participant received both the supplement and placebo at different times. The study found that the formulation increased whole-blood NAD⁺ in 26 participants over 28 days [13].
The evidence base is, however, limited to this single small study, and it carries a caveat that the design and funding of the paper were supported by the manufacturers of the formulation.
The systems biology framing is one of the more intellectually serious responses to the mechanistic gap in conventional precursor supplementation, but the clinical evidence is not yet sufficient to draw conclusions about functional outcomes.
Intravenous NAD⁺ is the most aggressively marketed option of all, offered by clinics on the basis that delivering NAD⁺ directly into the bloodstream bypasses the gut entirely and gets more into cells.
Owing to its size and charge, NAD⁺ cannot itself get directly into cells and has to be converted back into its precursors such as NMN, NR or NAM.
The theory is reasonable. The evidence is not.
A 2019 pilot study measured what actually happens to NAD⁺ in the blood and urine during a six-hour intravenous infusion [14]. Blood levels barely moved for the first two hours, and by six hours a large amount was appearing in the urine, consistent with rapid removal from the body and extracellular metabolism, with an uncertainty as to whether or how much reached the cells themselves.
A 2024 randomised trial directly comparing IV NAD⁺ against intravenous NR in healthy adults found that IV NAD⁺ caused moderate-to-severe nausea, raised heart rate, and chest pressure, while IV NR produced only minor tingling and raised NAD⁺ levels more effectively [15]. A 2026 real-world review confirmed the same pattern [16].
IV NAD⁺ also carries practical downsides that are rarely highlighted in clinic materials. The infusions are slow, averaging around 97 minutes, compared to 37 minutes for IV NR, because faster administration worsens the already common side effects of nausea, raised heart rate, and chest pressure.
There is a concern that standard IV infusion bags, typically made from polypropylene plastic, release an estimated 7,500 microplastic particles per litre of fluid directly into the bloodstream [17]. The long-term consequences of this are not yet known, but it is a non-trivial exposure from a route that currently lacks compelling evidence of benefit.
There is also an opportunity cost problem worth naming directly. The time, money, and effort required for a regular IV NAD⁺ protocol should, of course, not detract from sleep, structured exercise, and weight management. Three interventions with a vastly larger and more consistent evidence base for functional outcomes in ageing.
That does not make NAD⁺ biology irrelevant. But until the clinical evidence catches up with the biological rationale, the hierarchy of evidence still favours the foundations.
What the Clinical Trials Have Actually Shown
Three questions. Three different answers.
Question 1: Does oral NR or NMN raise blood NAD⁺ levels?
Yes, reliably. Multiple independent RCTs confirm that oral NR at 500–2,000 mg/day raises whole-blood NAD⁺ substantially above baseline, with dose-dependent increases of approximately 40–150% within two to four weeks [9], [10], [12]. This is not in dispute. It is also roughly the ceiling of what can be confidently concluded from most of the trial literature.
Question 2: Does raising blood NAD⁺ translate to improved function in healthy older adults?
Across meta-analyses, largely no.
A 2025 systematic review and meta-analysis pooled RCTs comparing NMN or NR against placebo in adults with a mean age above 60 and found no significant effects on skeletal muscle index, handgrip strength, or gait speed [18].
A separate 2024 meta-analysis of eight RCTs found no significant benefit of NMN on fasting glucose, fasting insulin, HbA1c, HOMA-IR, or lipid profiles in a generally healthy adult population [19].
Individual trials do show positive signals. A 12-week Japanese RCT in 42 healthy men aged 65 or over found improvements in gait speed and left-hand grip strength with NMN 250 mg/day [20], and a separate 12-week study found NMN maintained walking speed versus a placebo-group decline [21].
These, however, are not yet replicated when results are pooled in meta-analyses.
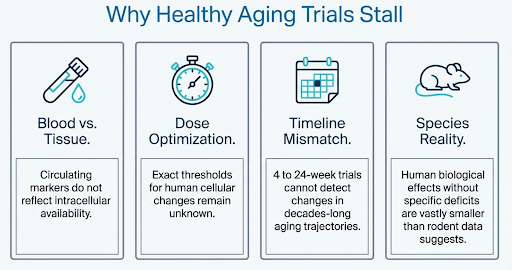
The deeper problem here is the surrogate marker used for NAD⁺ in many trials. Whole-blood NAD⁺ is a poor proxy for tissue NAD⁺. A value on a blood test is not the same as a significant change in levels inside the cells of the tissue you are trying to treat.
Question 3: Does NR or NMN improve outcomes in people with specific diseases where decreases in NAD⁺ are specifically implicated?
This is where the picture gets more interesting.
Peripheral artery disease. In a trial that randomised 90 patients to NR 1,000 mg/day or placebo for six months, NR improved six-minute walk distance by 31.0 metres in those patients who were adherent in their dosing [22]. This is the most rigorous functional outcome trial in this space but the benefit is in an arterial disease, not healthy ageing.
Parkinson's disease. Here a trial confirmed, for the first time in humans, that NR 1,000 mg/day raises brain NAD⁺ directly. Patients with the greatest increase showed improved energy metabolism and symptom scores [23]. A follow-up confirmed tolerability at 3,000 mg/day with further symptom improvement signs [24]. These are early stage findings only but sufficient to launch the next stage of dose-optimisation trials which are awaiting publication.
Heart failure. A 180-patient IV NAD⁺ trial in ischaemic cardiomyopathy reported improved cardiac function and symptom class, but lasted only seven days and needs replication [25].
Metabolic disease. NMN 250 mg/day for 10 weeks improved muscle insulin sensitivity in 25 postmenopausal women with prediabetes, measured by gold-standard clamp technique [11]. It did not lower blood glucose, improve lipids, reduce liver fat, or reduce inflammation.
The Central Problem
No robust randomised trial with hard endpoints that matter - mortality, hospitalisation, major cardiac events, dementia - has been completed in this field. The evidence base is almost entirely pilot studies, early (phase I and II) trials, and surrogate markers.
Most trials run for four to twenty-four weeks. That is almost certainly insufficient to detect effects on trajectories that unfold over years.
The blood NAD⁺ surrogate marker is not entirely convincing. When blood NAD⁺ rises while the real outcomes that matter remain largely unchanged in healthy older adults, there are several possible explanations:
blood is not a reliable proxy for target tissue;
duration and dose are not yet optimised;
functional outcomes need longer timelines to shift; or
the biological effect of raising NAD⁺ in well-nourished older adults without a specific disease-related NAD⁺ deficit is simply smaller than the rodent data suggested.
Probably some combination of all four.
Some researchers have highlighted that a consistent age-related decline in NAD⁺ levels is not equal among tissues and is seen in muscle but not in blood. However, NAD⁺ boosting doesn’t seem to change muscle NAD⁺ levels, while easily increasing blood NAD⁺ concentration [26].
There is an additional, less-discussed complication that results from taking a whole-systems approach to NAD⁺ supplementation that maybe touches on points 1 & 4 above.
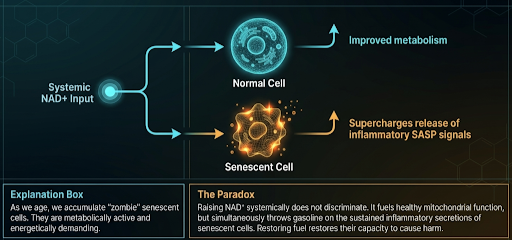
As we age, a growing proportion of our cells enter a state called senescence, or what some have called “zombie cells”. These are cells that have permanently stopped dividing, usually in response to accumulated DNA damage or stress, and rather than dying, they survive in the body and release a sustained stream of inflammatory signals to their neighbouring cells.
In youth, the immune system removes them efficiently; with age, they accumulate. They are not inert or benign. They are metabolically active. The ability to maintain that inflammatory output is energetically demanding, and thus requires NAD⁺.
This creates a problem for NAD⁺ supplementation that is not widely acknowledged. Raising NAD⁺ levels systemically does not selectively target healthy cells. It raises NAD⁺ availability throughout the body, including in senescent cells, which then have more fuel to sustain and amplify their inflammatory secretions.
Thus restoring NAD⁺ may in part be restoring the capacity of senescent cells to cause harm [4].
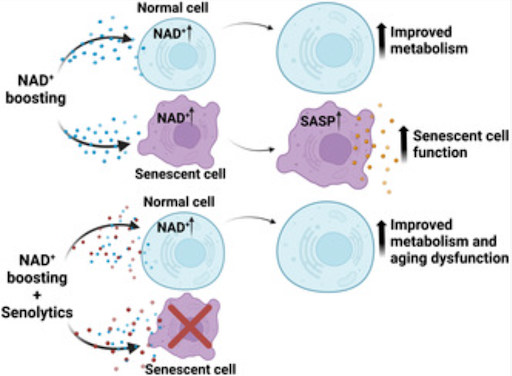
Experimental preclinical evidence shows that NMN supplementation can amplify the inflammatory signals of senescent cells rather than suppress it, with NMN supplementation enhancing this activity across multiple senescence models and accelerating cancer progression in mouse models enriched with senescent cells [27].
The net effect is that NAD⁺ repletion may produce two simultaneous and opposing outcomes: improved mitochondrial function in healthy cells on one side, and amplified inflammatory signalling from senescent cells on the other.
Which effect dominates, and in which tissue, at which dose, in which individual, is not known. What this suggests is that for NAD⁺ supplementation to work as intended, the burden of senescent cells may first need to be addressed. Giving more fuel to a tissue where senescent cells are already abundant may be less effective, and potentially counterproductive, compared to clearing those cells first and then restoring NAD⁺. That is still an unknown and not been tested in humans at scale.
Source: [4]
The bigger picture is that NAD⁺ decline is a real feature of ageing biology.
The mechanisms are well characterised. But NAD⁺ sits inside a whole system - one involving inflammatory tone, mitochondrial quality control, DNA damage load, and metabolic function - and supplementing one precursor input addresses only part of that system.
The trials where the clearest signals emerge are the ones targeting populations where that system is already significantly disrupted: PAD, Parkinson's, heart failure. That is worth remembering when evaluating claims made to healthy people in the absence of a clear deficit in their NAD⁺ levels.
Bottom line
You can raise blood NAD+ in humans.
You cannot currently claim that this produces reliable, clinically meaningful benefit in most populations.
Today, the most realistic stance is that NAD+ precursors are biologically active, clinically unproven, and overinterpreted.
That may change.
But the field needs better tissue data, better outcome data, and longer trials before the claims catch up with the marketing.
Q&A
Should people who are currently taking NR or NMN stop?
That is a clinical decision involving context this piece cannot cover. What I would say the evidence supports: these compounds are safe at standard doses, reliably raise blood NAD⁺, and in populations with specific conditions, there are encouraging signals.
In healthy older adults without those conditions, the case for meaningful functional benefit from general ageing supplementation has not yet been established.
Is IV NAD⁺ worthwhile compared to oral supplementation?
On current evidence, no.
IV NAD⁺ has worse tolerability than IV NR, uncertain tissue delivery, and no published randomised trial data on functional outcomes. I would want to see considerably more rigorous data before recommending it over an oral precursor.
What would actually move the evidence forward?
A powered, multi-year randomised, controlled trial in with pre-specified hard clinical endpoints. Until those trials exist, the honest answer to "does this work?" remains: we know it raises a blood marker, there are promising signals in specific clinical contexts, and the hard evidence is not yet there.
Further Reading
[1] J. R. Revollo, A. A. Grimm, and S. Imai, ‘The regulation of nicotinamide adenine dinucleotide biosynthesis by Nampt/PBEF/visfatin in mammals’, Curr. Opin. Gastroenterol., vol. 23, no. 2, pp. 164–170, Mar. 2007, doi: 10.1097/MOG.0b013e32801b3c8f.
[2] J. Yoshino, K. F. Mills, M. J. Yoon, and S. Imai, ‘Nicotinamide Mononucleotide, a Key NAD+ Intermediate, Treats the Pathophysiology of Diet- and Age-Induced Diabetes in Mice’, Cell Metab., vol. 14, no. 4, pp. 528–536, Oct. 2011, doi: 10.1016/j.cmet.2011.08.014.
[3] J. Camacho-Pereira et al., ‘CD38 Dictates Age-Related NAD Decline and Mitochondrial Dysfunction through an SIRT3-Dependent Mechanism’, Cell Metab., vol. 23, no. 6, pp. 1127–1139, Jun. 2016, doi: 10.1016/j.cmet.2016.05.006.
[4] C. C. S. Chini, H. S. Cordeiro, N. L. K. Tran, and E. N. Chini, ‘NAD metabolism: Role in senescence regulation and aging’, Aging Cell, vol. 23, no. 1, p. e13920, Jul. 2023, doi: 10.1111/acel.13920.
[5] E. D’Andrea, S. P. Hey, C. L. Ramirez, and A. S. Kesselheim, ‘Assessment of the Role of Niacin in Managing Cardiovascular Disease Outcomes: A Systematic Review and Meta-analysis’, JAMA Netw. Open, vol. 2, no. 4, p. e192224, Apr. 2019, doi: 10.1001/jamanetworkopen.2019.2224.
[6] E. S. Hwang and S. B. Song, ‘Possible Adverse Effects of High-Dose Nicotinamide: Mechanisms and Safety Assessment’, Biomolecules, vol. 10, no. 5, p. 687, May 2020, doi: 10.3390/biom10050687.
[7] N. J. Connell et al., ‘NAD+-Precursor Supplementation With L-Tryptophan, Nicotinic Acid, and Nicotinamide Does Not Affect Mitochondrial Function or Skeletal Muscle Function in Physically Compromised Older Adults’, J. Nutr., vol. 151, no. 10, pp. 2917–2931, Oct. 2021, doi: 10.1093/jn/nxab193.
[8] S. A. J. Trammell et al., ‘Nicotinamide riboside is uniquely and orally bioavailable in mice and humans’, Nat. Commun., vol. 7, no. 1, p. 12948, Oct. 2016, doi: 10.1038/ncomms12948.
[9] C. R. Martens et al., ‘Chronic nicotinamide riboside supplementation is well-tolerated and elevates NAD+ in healthy middle-aged and older adults’, Nat. Commun., vol. 9, no. 1, p. 1286, Mar. 2018, doi: 10.1038/s41467-018-03421-7.
[10] S. Christen et al., ‘The differential impact of three different NAD+ boosters on circulatory NAD and microbial metabolism in humans’, Nat. Metab., vol. 8, no. 1, pp. 62–73, Jan. 2026, doi: 10.1038/s42255-025-01421-8.
[11] M. Yoshino et al., ‘Nicotinamide mononucleotide increases muscle insulin sensitivity in prediabetic women’, Science, vol. 372, no. 6547, pp. 1224–1229, Jun. 2021, doi: 10.1126/science.abe9985.
[12] K. M. Pencina et al., ‘MIB-626, an Oral Formulation of a Microcrystalline Unique Polymorph of β-Nicotinamide Mononucleotide, Increases Circulating Nicotinamide Adenine Dinucleotide and its Metabolome in Middle-Aged and Older Adults’, J. Gerontol. Ser. A, vol. 78, no. 1, pp. 90–96, Jan. 2023, doi: 10.1093/gerona/glac049.
[13] J. D. Henderson, S. N. Z. Quigley, S. S. Chachra, N. Conlon, and D. Ford, ‘The use of a systems approach to increase NAD+ in human participants’, Npj Aging, vol. 10, no. 1, p. 7, Feb. 2024, doi: 10.1038/s41514-023-00134-0.
[14] R. Grant et al., ‘A Pilot Study Investigating Changes in the Human Plasma and Urine NAD+ Metabolome During a 6 Hour Intravenous Infusion of NAD+’, Front. Aging Neurosci., vol. 11, Sep. 2019, doi: 10.3389/fnagi.2019.00257.
[15] J. Hawkins et al., ‘Randomized, placebo-controlled, pilot clinical study evaluating acute Niagen®+ IV and NAD+ IV in healthy adults’, Jun. 10, 2024, medRxiv. doi: 10.1101/2024.06.06.24308565.
[16] K. Reyna et al., ‘Intravenous infusion of nicotinamide adenine dinucleotide (NAD+) versus nicotinamide riboside (NR): a retrospective tolerability pilot study in a real-world setting’, Front. Aging, vol. 7, Feb. 2026, doi: 10.3389/fragi.2026.1652582.
[17] T. Huang et al., ‘MPs Entering Human Circulation through Infusions: A Significant Pathway and Health Concern’, Environ. Health, vol. 3, no. 5, pp. 551–559, Feb. 2025, doi: 10.1021/envhealth.4c00210.
[18] K. Prokopidis, F. Moriarty, G. Bahat, J. McLean, D. D. Church, and H. P. Patel, ‘The Effect of Nicotinamide Mononucleotide and Riboside on Skeletal Muscle Mass and Function: A Systematic Review and Meta-Analysis’, J. Cachexia Sarcopenia Muscle, vol. 16, no. 3, p. e13799, Jun. 2025, doi: 10.1002/jcsm.13799.
[19] J. Zhang, E. T.-C. Poon, and S. H.-S. Wong, ‘Efficacy of oral nicotinamide mononucleotide supplementation on glucose and lipid metabolism for adults: a systematic review with meta-analysis on randomized controlled trials’, Crit. Rev. Food Sci. Nutr., vol. 65, no. 22, pp. 4382–4400, Aug. 2025, doi: 10.1080/10408398.2024.2387324.
[20] M. Igarashi et al., ‘Chronic nicotinamide mononucleotide supplementation elevates blood nicotinamide adenine dinucleotide levels and alters muscle function in healthy older men’, Npj Aging, vol. 8, no. 1, p. 5, May 2022, doi: 10.1038/s41514-022-00084-z.
[21] M. Morifuji, S. Higashi, S. Ebihara, and M. Nagata, ‘Ingestion of β-nicotinamide mononucleotide increased blood NAD levels, maintained walking speed, and improved sleep quality in older adults in a double-blind randomized, placebo-controlled study’, GeroScience, vol. 46, no. 5, pp. 4671–4688, Oct. 2024, doi: 10.1007/s11357-024-01204-1.
[22] M. M. McDermott et al., ‘Nicotinamide riboside for peripheral artery disease: the NICE randomized clinical trial’, Nat. Commun., vol. 15, no. 1, p. 5046, Jun. 2024, doi: 10.1038/s41467-024-49092-5.
[23] B. Brakedal et al., ‘The NADPARK study: A randomized phase I trial of nicotinamide riboside supplementation in Parkinson’s disease’, Cell Metab., vol. 34, no. 3, pp. 396-407.e6, Mar. 2022, doi: 10.1016/j.cmet.2022.02.001.
[24] H. Berven et al., ‘NR-SAFE: a randomized, double-blind safety trial of high dose nicotinamide riboside in Parkinson’s disease’, Nat. Commun., vol. 14, no. 1, p. 7793, Nov. 2023, doi: 10.1038/s41467-023-43514-6.
[25] ‘Effect of Nicotinamide Adenine Dinucleotide on Heart Failure Caused by Ischemic Cardiomyopathy: A Randomized, Placebo-Controlled Trial | American Journal of Cardiovascular Drugs | Springer Nature Link’. Accessed: Apr. 10, 2026. [Online]. Available: https://link.springer.com/article/10.1007/s40256-025-00764-7
[26] K. T. Vinten et al., ‘NAD+ precursor supplementation in human ageing: clinical evidence and challenges’, Nat. Metab., vol. 7, no. 10, pp. 1974–1990, Oct. 2025, doi: 10.1038/s42255-025-01387-7.
[27]T. Nacarelli et al., ‘NAD+ metabolism governs the proinflammatory senescence-associated secretome’, Nat. Cell Biol., vol. 21, no. 3, pp. 397–407, Mar. 2019, doi: 10.1038/s41556-019-0287-4.