Part 1. ApoB & Preventing Heart Disease. Your cholesterol looks fine. Are you sure?
Many people are familiar with the concept that high cholesterol is a significant risk factor for heart disease, and even that LDL-cholesterol levels in the blood give a good reflection of this risk.
The truth is that another particle in the blood, Apolipoprotein B (ApoB), is closer to the underlying biology of atherosclerosis than LDL-cholesterol (LDL-C).
Some therefore argue we should be aggressively targeting lower levels of apoB. The real question, in a healthy population, is where earlier intervention is supported, where it is still inference, and where current prevention guidelines remain too reactive.
It’s a familiar line
"Your cholesterol is fine."
Cardiovascular disease still kills more people than any other condition worldwide, and much of that risk develops quietly over decades, which is exactly why it matters that we may still be measuring risk too crudely and acting too late [1].
LDL-cholesterol became central for a good reason. It measures the cholesterol carried in the particles most strongly implicated in atherosclerosis, and lowering it has clearly reduced cardiovascular events [2].
So when people hear, “your cholesterol is fine”, it usually means LDL-cholesterol is not especially high.
The problem is not that LDL-C doesn’t matter. It is that it’s often treated as the main decision trigger, rather than one imperfect proxy within a larger system of cardiovascular risks.
Apolipoprotein B
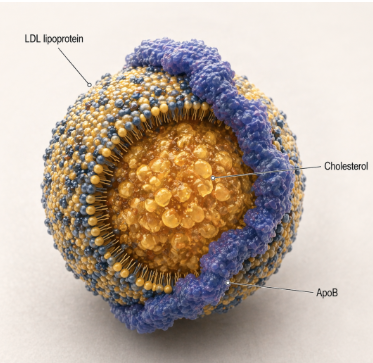
LDL-C tells us how much cholesterol is being carried in the bloodstream by LDL particles, regardless of how many particles there are.
ApoB tells us how many potentially harmful lipoprotein particles are circulating, carrying the cholesterol.
Those are not the same thing. One crude analogy goes like this: LDL-C tells us how much cargo is being moved. ApoB tells us how many lorries are on the road carrying it.
You can move the same total load in a small number of heavily loaded lorries, or in a much larger number of lightly loaded ones.
When it comes to the arteries, the number of vehicles matters, because each passing particle is another chance for one to enter and get trapped in the vessel wall, causing narrowing and reduced blood flow to the heart muscle.
A person can be carrying a modest amount of cholesterol per particle, but still have a high number of particles passing through the arteries. In that situation, particle number may be the more important question [3].
Using one standard cholesterol measure and a 10 year risk calculator is a reasonable starting point for population level prevention.
But it can miss people whose real burden of harmful cholesterol carrying particles is higher than their LDL-C alone suggests.
It can also delay action, because atherosclerosis develops through years of cumulative exposure, not just when a shorter term (10 year) risk score finally becomes high enough to trigger treatment.
So the real question is not whether apoB is "better". It is whether our current model is too reactive because it confuses a proxy with the burden that actually drives disease.
What apoB measures, and why that matters
Atherosclerosis begins when apoB-containing particles enter and are retained in the arterial wall.
Each atherogenic LDL particle, loaded with cholesterol, carries one apoB molecule.
The distinction from LDL-C matters most when the cholesterol content of each LDL particle is low, because LDL-C levels can look fairly normal even when the number of particles is high.
This is common in insulin resistance, hypertriglyceridaemia, central adiposity, and type 2 diabetes [4], [5]. In those settings, LDL-C results can look reasonable while the particle number is actually quite high.
What the human evidence actually shows
Discordance studies. When apoB and LDL-C disagree, apoB usually predicts risk more closely. The Marston analysis across UK Biobank, FOURIER, and IMPROVE-IT is one of the clearest examples. Across primary (preventing the first event) and secondary (preventing another event) prevention of heart disease, apoB remained the more consistent marker of risk of heart attacks [6].
That does not prove that targeting apoB in treatment will always outperform targeting LDL-C. It does show that apoB is often the better summary of the cardiovascular risk we are trying to estimate. Currently, there is no study to date comparing the benefits of apoB guided treatment over LDL-C guided treatment.
Mendelian randomisation. This is where the evidence moves beyond prediction.
Mendelian randomisation is useful here because it helps separate correlation from causation, using inherited genetic differences as a kind of natural lifelong trial.
Mendelian randomisation is not a clinical trial. It does not tell us directly what happens if a healthy 40-year-old starts therapy today.
What it does do is strengthen our ability to infer a causal effect over the life course. In a 2020 Mendelian randomisation analysis, apoB emerged as the dominant causal risk signal for coronary heart disease once LDL-C and triglycerides were accounted for. That matters because it suggests particle number is not just a better correlate. It is closer to the thing driving disease [7].
That is stronger than a mechanism. It is still not the same as a study showing that a specific apoB threshold should trigger treatment in all healthy adults, yet.
Earlier exposure reduction. This is the part of the argument that is directionally right but can be phrased prematurely strongly. The thought process on the current weight of evidence today goes: if atherogenic particle burden (apoB level) is causal, and cumulative exposure (how long you have high levels of apoB) matters, then reducing that exposure earlier (ie lowering apoB) should plausibly reduce lifetime risk more than waiting until a 10 year risk calculator threshold.
That proposition is supported by genetics, by outcome trials of lipid lowering more broadly, and now by guidelines using language that explicitly leans toward earlier intervention to reduce lifelong risk from prolonged exposure to atherogenic lipoproteins [8].
Where apoB helps now
This is where the interpretation needs to stay disciplined. Not everyone needs earlier drug therapy. Not every healthy adult needs an aggressive apoB target. But apoB is already useful where standard framing is most likely to understate the risk burden.
In metabolic dysfunction: insulin resistance, raised triglycerides, type 2 diabetes, central adiposity, LDL-C can look more reassuring than the underlying apoB levels would suggest [8], [9].
In younger adults whose cardiovascular risk is accumulating faster than it should: the important question here is less the probability of a heart attack today, but more the cumulative burden over the next two or three decades, or longer.
In those where there is apparent control with residual concern: LDL-C appears to be "at goal" but the overall picture still has features suggesting residual cardiovascular risk. In that situation, apoB can help identify whether atherogenic particle levels remain elevated despite apparently satisfactory standard lipids.
In imaging–risk mismatch: arteries that look older than the lipid panel suggests. The panel may not be telling the whole story. ApoB can be part of that clarification, although imaging-guided treatment strategies are still not the same as apoB-target trials.
Bottom Line
ApoB and LDL-C are not the same biological object. LDL-C measures cholesterol mass inside LDL particles. ApoB counts the atherogenic particles themselves.
When the two diverge, most often in metabolic dysfunction, apoB is the more consistent marker of cardiovascular risk. Genetic evidence from Mendelian randomisation suggests particle number is closer to the causal driver of atherosclerosis than cholesterol mass, not just a better correlate.
That reframes, rather than replaces, the standard lipid conversation. A "normal" LDL-C or a low 10-year risk score is not the same as a quiet biology — especially in people whose particles are cholesterol-depleted, whose risk trajectory is unfavourable, or whose imaging tells a different story from the panel.
How aggressively apoB should be lowered, and in whom, is the subject of Part 2.
Q&A
If LDL-C is only a surrogate, can I ignore my LDL-C result? No. LDL-C still matters operationally. Most of the treatment evidence and most clinical pathways were built around it, and in many people it remains a reasonable summary of cardiovascular risk. The point is not to replace it, but to recognise where it is most likely to mislead. ApoB adds information when the two diverge.
Should I ask for an apoB test at my next lipid check? It is reasonable in specific situations: raised triglycerides, insulin resistance or type 2 diabetes, central adiposity, a strong family history of early cardiovascular disease, or when the lipid panel looks reassuring but the clinical picture does not. It is not yet a test every healthy adult needs routinely, though the 2026 guideline broadens the circumstances in which it is recommended.
If apoB is biologically closer to the driver, why isn't it the default test? Partly because much of the trial evidence and many clinical pathways were built around LDL-C. Partly because apoB availability, laboratory reporting, and reimbursement still vary. And partly because agreed interpretive frameworks and thresholds are still evolving. Guidelines are moving in the direction of broader use, but they have not yet made apoB the primary default marker.
What does "discordance" between apoB and LDL-C actually mean? It means the two markers are pointing at different risk categories. Most often, particularly in metabolic dysfunction, LDL-C looks more reassuring than apoB, because the same amount of cholesterol is distributed across a higher number of smaller, cholesterol-depleted particles. When that happens, apoB gives a more accurate estimate of atherogenic burden than LDL-C does.
Does a high apoB automatically mean I need treatment? No. ApoB is a risk marker, not a treatment trigger on its own. It should be interpreted alongside age, risk trajectory, family history, metabolic phenotype, and, where relevant, imaging. How aggressively to act on a raised apoB is a separate question, and one the evidence handles less cleanly than the measurement question itself. Part 2 takes that up directly.
Further Reading
[1] ‘Cardiovascular diseases (CVDs)’. Accessed: Apr. 17, 2026. [Online]. Available: https://www.who.int/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds)
[2] Cholesterol Treatment Trialists’ (CTT) Collaboration et al., ‘Efficacy and safety of more intensive lowering of LDL cholesterol: a meta-analysis of data from 170,000 participants in 26 randomised trials’, Lancet, vol. 376, no. 9753, pp. 1670–1681, Nov. 2010, doi: 10.1016/S0140-6736(10)61350-5.
[3] ‘Physiological Bases for the Superiority of Apolipoprotein B Over Low‐Density Lipoprotein Cholesterol and Non–High‐Density Lipoprotein Cholesterol as a Marker of Cardiovascular Risk | Journal of the American Heart Association’. Accessed: Apr. 23, 2026. [Online]. Available: https://www.ahajournals.org/doi/10.1161/JAHA.122.025858
[4] O. P. Ganda, C. G. Jumes, M. J. Abrahamson, and M. Molla, ‘Quantification of concordance and discordance between apolipoprotein-B and the currently recommended non-HDL-cholesterol goals for cardiovascular risk assessment in patients with diabetes and hypertriglyceridemia’, Diabetes Res. Clin. Pract., vol. 97, no. 1, pp. 51–56, Jul. 2012, doi: 10.1016/j.diabres.2012.02.013.
[5] S. A. Varvel et al., ‘Discordance between apolipoprotein B and low-density lipoprotein particle number is associated with insulin resistance in clinical practice’, J. Clin. Lipidol., vol. 9, no. 2, pp. 247–255, 2015, doi: 10.1016/j.jacl.2014.11.005.
[6] N. A. Marston et al., ‘Association of Apolipoprotein B–Containing Lipoproteins and Risk of Myocardial Infarction in Individuals With and Without Atherosclerosis: Distinguishing Between Particle Concentration, Type, and Content’, JAMA Cardiol., vol. 7, no. 3, pp. 250–256, Mar. 2022, doi: 10.1001/jamacardio.2021.5083.
[7] T. G. Richardson et al., ‘Evaluating the relationship between circulating lipoprotein lipids and apolipoproteins with risk of coronary heart disease: A multivariable Mendelian randomisation analysis’, PLoS Med., vol. 17, no. 3, p. e1003062, Mar. 2020, doi: 10.1371/journal.pmed.1003062.
[8] Writing Committee Members et al., ‘2026 ACC/AHA/AACVPR/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Dyslipidemia: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines’, Circulation, vol. 0, no. 0, doi: 10.1161/CIR.0000000000001423.
[9] D. E. Soffer et al., ‘Role of apolipoprotein B in the clinical management of cardiovascular risk in adults: An Expert Clinical Consensus from the National Lipid Association’, J. Clin. Lipidol., vol. 18, no. 5, pp. e647–e663, Sep. 2024, doi: 10.1016/j.jacl.2024.08.013.