From pre to post menopause: how we underestimate cardiovascular risk in women
The cardiovascular risk shift from pre to post menopause
Middle-aged women have the fastest relative increase in death from atherosclerotic heart disease [1].
For many women, the problem is not that cardiovascular risk suddenly increases after menopause.
It is that we apply a cardiovascular risk assessment framework used before the menopause, that is suboptimal to capture what is unfolding through the transition to a post-menopausal state.
A woman can be lean, active, metabolically healthier than average, and far more health-literate than most of the population, but that still doesn’t ensure that her cardiovascular risk is being well interpreted.
This is important because there are a whole range of changes that occur around the menopause that mean that this is much more than a period of symptom management alone.
It is also a vascular and metabolic transition, with changes in blood pressure, adiposity (weight change), cholesterol, insulin sensitivity, and blood vessel function that can make the usual cardiovascular risk models look more reassuring [2].
The usual tools we use are not wrong, they are just incomplete and inadequate to capture the physiology specific to women around the menopause.
Premenopausal oestrogen effects can be favourable, but they do not erase lifetime exposure to apoB containing particles, blood pressure, inflammation, and inherited risk.
A low 10 year score can coexist with meaningful lifetime risk.
It’s not that this is wrong but it is just not always enough. The European Atherosclerosis Society call to action on women, lipids, and ASCVD makes the broader point that cardiovascular risk in women is often underappreciated, diagnosis is delayed, and treatment is often less than optimal [1].
Has the old model had its day?
Cardiovascular prevention has long been shaped by data, thresholds, and disease models that fit men far better than women. Sex-specific risk factors, cholesterol changes across different life stages, and female-predominant phenotypes have rarely been integrated well into routine prevention considerations.
The result is not that clinicians know nothing about cardiovascular risk in women.
It is that the default risk assessment can be too crude, especially around menopause.
Before menopause, short-term event risk in many women is low. But short-term risk and lifetime risk are not the same thing.
If atherosclerotic disease develops over decades, then a 10-year calculator can miss a meaningful part of the picture in a woman moving through her midlife, especially when biomarkers, family history, inflammation, or symptoms do not fit the picture.
The Women’s Health Study analysis of 30-year cardiovascular found that in initially healthy women, baseline hsCRP (a marker of inflammation), LDL-cholesterol, and Lp(a) (a largely genetically-determined marker of cardiovascular risk) each predicted long-term cardiovascular events over three decades [3].
What actually changes across the transition
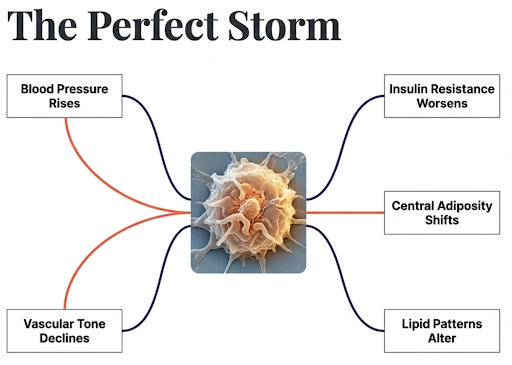
The menopause transition is associated with several cardiometabolic changes that tend to move in the wrong direction at once [4].
Blood pressure tends to rise.
Body fat distribution often shifts centrally.
Insulin resistance can worsen.
Lipid patterns often become less favourable.
Endothelial and vascular (blood vessel) changes also become significant.
This doesn’t mean every woman suddenly becomes high risk around the menopause. It does mean this is a period where a reassessment is worthwhile. Trends matter. Context matters.
A better way to think about this is that the menopause itself is a window in which risk can accelerate, conventional markers can become less informative in isolation, and the threshold for a broader review may drop.
Why LDL-cholesterol alone is often not enough
LDL-C still matters. It remains a useful marker and a causal pathway in atherosclerotic disease. But LDL-C is not the whole story, especially when the clinical picture feels off.
ApoB can add clarity because it reflects the number of atherogenic particles, not only the cholesterol mass inside them.
In cases where we see a mismatch, LDL-C can look acceptable while the particle number (conferred by ApoB) is high.
Using ApoB can resolve that mismatch. It is not a replacement for clinical judgement but adds colour to the picture [1].
Lp(a) also deserves more attention, but for a different reason. Lp(a) is largely inherited.
Lifestyle rarely shifts it meaningfully. That matters because a person can look low risk on standard lipids, yet carry higher lifetime atherothrombotic risk.
Measuring it once can prevent years of false reassurance.
hsCRP denotes inflammatory levels. It is not a specific cardiovascular marker or a marker for a specific diagnosis. It adds insight, in the right context, to signal that the low-risk assessment might not be quite so certain. It refines judgement, rather than replacing it.
We need better stratification across the menopause, and it makes little sense to rely on LDL-C alone and ignore ApoB, Lp(a), inflammation, and the direction of change over time.
Women are not men!
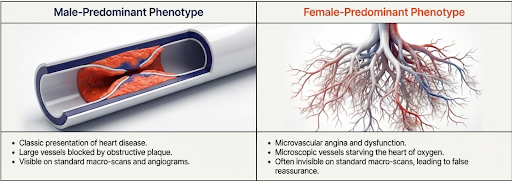
Jokes aside, this issue is important because women do not always present like men with obstructive coronary disease.
Microvascular angina and coronary dysfunction without obstructive disease appear more common in women than in men. This is due to small vessels being blocked, rather than the larger ones.
A normal angiogram (scan of the arteries to the heart) can rule out large vessel blockage and still leave symptoms unexplained. A specialised heart test called stress testing may be abnormal despite what would look like normal large blood vessels on a normal angiogram.
That is a reason to avoid early reassurance when symptoms persist. A woman can have genuine symptoms due to a lack of blood flow to the heart, without the kind of large-vessel, stereotypical obstructive plaque that many people still imagine causes heart attacks [5].
That does not mean every symptomatic woman with a normal angiogram has microvascular dysfunction. It does mean that a normal angiogram is not always the end of the story though and needs careful consideration. This is one reason women are sometimes reassured too early.
Why lifetime risk matters
As we noted above, in the Women’s Health Study analysis, long-term follow-up showed that baseline hsCRP, LDL-C, and Lp(a) each signaled increased risk for future cardiovascular events over 30 years in initially healthy women [3]. The message is not that every woman needs more tests but that a narrow 10-year cardiovascular risk calculator can understate cumulative exposure and lifetime burden in a group where the disease process accelerates often at a later date.
This is the piece that is especially relevant during the menopause transition. This is the period when often body composition shifts, blood pressure rises, lipids worsen, symptoms change, and the gap between apparent health (existing risk calculators) and actual (lifetime risk and additional biomarker considerations) risk classification can widen.
So we should be asking ourselves not just, what is the 10-year risk today, but what assumptions might we be making and what are we overlooking?
What better risk stratification looks like
This is not arguing for indiscriminate testing. It is a case for better judgement.
This means a detailed family history, Lp(a) measured at least once and an ApoB when the phenotype and LDL-C do not match.
And it helps to think in terms of lifetime rather than purely 10-year terms, especially in women with strong family history, metabolic dysfunction, inflammation or other risk factors.
During peri-menopause, the emphasis should shift towards the importance of trend more than single thresholds. A woman whose lipids, waist circumference, blood pressure, or glucose control, are seeing step changes should not be interpreted through the same lens as five years earlier.
After menopause, broader review becomes more important.
Short-term calculators can still be useful but they are just not enough on their own. Additional risk factors add value. Alongside Lp(a), ApoB and hsCRP, coronary artery calcium scoring can be considered selectively for risk refinement when the conventional estimate feels incomplete or falsely reassuring. This is a risk-refinement, not a blanket screening recommendation.
Key Takeways
Treat menopause as a cardiometabolic risk transition, not only a symptom transition.
Use trend data. A step change in waist, blood pressure, glucose, or lipids matters more than a single threshold.
Do not rely on LDL C alone in discordant cases. Consider ApoB and Lp(a) for risk refinement.
Do not use hsCRP as a diagnosis. Use it as context when the picture feels falsely reassuring.
Keep on top of the foundations: Blood pressure and blood sugar control, resistance training, cardiorespiratory fitness, good body composition, sleep, and smoking avoidance drive the biggest absolute risk reduction.
Q&A
Should every woman have Lp(a) measured?
Measuring it at least once is increasingly useful for long-term risk stratification, especially when family history is strong or the overall risk picture feels discordant.
Does a normal angiogram rule out a coronary problem?
No. It rules out certain types of obstructive disease more than it rules out all clinically relevant coronary dysfunction. In women, microvascular angina is more common and stress testing can be abnormal despite a normal angiogram.
Should menopause automatically trigger statin therapy?
No. Menopause should trigger better interpretation of overall risk, including risk factors, biomarker discordance, symptoms, and lifetime burden. Treatment decisions still need to be individualised.
Further reading
[1] ‘Women, lipids, and atherosclerotic cardiovascular disease: a call to action from the European Atherosclerosis Society | European Heart Journal | Oxford Academic’. Accessed: Apr. 01, 2026. [Online]. Available: https://academic.oup.com/eurheartj/article/44/39/4157/7249105
[2] S. R. El Khoudary et al., ‘Menopause Transition and Cardiovascular Disease Risk: Implications for Timing of Early Prevention: A Scientific Statement From the American Heart Association’, Circulation, vol. 142, no. 25, pp. e506–e532, Dec. 2020, doi: 10.1161/CIR.0000000000000912.
[3] P. M. Ridker, M. V. Moorthy, N. R. Cook, N. Rifai, I.-M. Lee, and J. E. Buring, ‘Inflammation, Cholesterol, Lipoprotein(a), and 30-Year Cardiovascular Outcomes in Women’, N. Engl. J. Med., vol. 391, no. 22, pp. 2087–2097, Dec. 2024, doi: 10.1056/NEJMoa2405182.
[4] ‘Menopause Transition and Cardiovascular Disease Risk: Implications for Timing of Early Prevention: A Scientific Statement From the American Heart Association’, Circulation, Accessed: Apr. 01, 2026. [Online]. Available: https://www.ahajournals.org/doi/10.1161/CIR.0000000000000912
[5] U. Tayal et al., ‘Advancing the access to cardiovascular diagnosis and treatment among women with cardiovascular disease: a joint British Cardiovascular Societies’ consensus document’, Nov. 2024, doi: 10.1136/heartjnl-2024-324625.