Part 2. ApoB & Preventing Heart Disease. How low can you go?
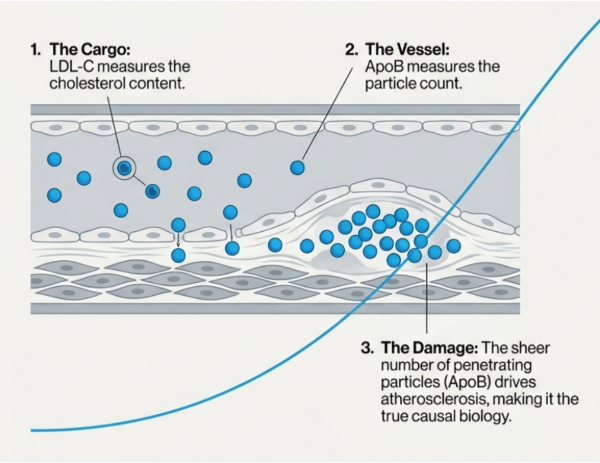
There is a strong case that apoB is biologically closer to the driver of atherosclerosis than LDL-C [1], [2].
There is a strong case that discordance matters.
There is a reasonable case that current prevention often starts too late.
There is not yet a direct evidence base for a universal strategy of treating all healthy adults to apoB below 0.6 g/L or lower.
That threshold may look biologically attractive [3]. It may prove useful in some individuals but it remains an extrapolated and nuanced position, not yet one settled with robust evidence.
Why? Because the evidence is incomplete. We have strong biology. We have strong causal inference from genetics [3]. We have strong data in discordant populations [4], [5]. We have strong trial evidence that lowering atherogenic lipoproteins reduces events [6], [7].
What we do not have is a dedicated primary prevention trial showing that treating healthy adults to apoB below, for example 0.6 g/L produces better health outcomes than current practice.
That absence does not make the idea wrong. It does mean the confidence attached to it should be a nuanced discussion between clinician and patient.
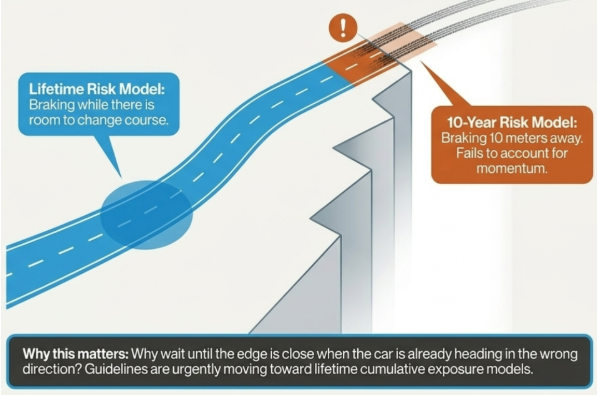
Why short-term risk calculators often miss the real issue
A 10-year calculator is useful for estimating near-term probability of a heart attack or stroke. It is not a full analysis of a lifetime biological burden. And it is particularly limited in younger adults, because age heavily impacts the result.
So someone can have a strong family history, metabolic dysfunction, discordantly high apoB, early scans suggesting disease, and a high risk cumulative exposure picture and still look "low risk" in a short-term model due to their age.
That does not make the calculator useless. It means the calculator is answering a shorter term question than many clinicians and patients are now asking. This is why guidelines are moving towards lifetime risk calculators, personalisation of risk assessment, including the use of apoB and imaging scans as additional ways to individualise discussions around cardiovascular risks [8].
One way to think about the difference between 10-year and lifetime risk calculators is this: imagine you are driving towards a cliff edge. A 10 year risk calculator is a bit like telling you to brake when you are 10 metres away and hoping that is early enough. A lifetime risk model asks a better question. Why wait until the edge is close, when you already know the car is heading in the wrong direction? It is about braking earlier, while there is still more room to change course.
We can get cholesterol low but the real question is “what are the costs of getting it there?”
There are really two different safety questions.
The first is pathological: does having very low levels of LDL-C or apoB itself appear harmful?
The second is pharmacological: what side effects, treatment burden, and long-term trade-offs come with using medication to push levels that low?
On the pathological side, the current evidence is broadly reassuring. Contemporary trial data in high risk patient groups do not show convincing evidence that intensive lipid lowering causes major neurocognitive harm, dementia, or a clear excess of haemorrhagic stroke in selected patients, even at very low achieved LDL-C levels [9], [10], [11], [12], [13], [14], [15], [16].
The current significant caveat is that most of these data come from people at high or very high cardiovascular risk, followed for a moderate number of years. They do not yet fully answer the question we’d like: what is the net effect of driving LDL-C or apoB to low levels for decades in otherwise healthy younger adults?
So if, even in the frailest populations, there are not strong signals of pathological harm, the real concern shifts toward pharmacology and side effects.
If you try to push apoB below 0.6 g/L across a broad healthy population, you are not mainly testing whether low apoB is biologically dangerous. You are testing whether the medications, side effects, costs, adherence burden (how many pills you cumulatively have to pop each day), and long-term medicalisation are justified by the likely absolute benefit.
For statins, that means muscle symptoms, a small excess risk of diabetes in susceptible individuals, and the practical reality that long-term adherence in primary prevention is often imperfect. For non-statin therapy, it means adding cost, complexity, injections in some cases, and a thinner evidence base for decades of use in low-risk people. The closer someone is to genuinely high lifetime risk, the more those trade-offs may be worth making. The lower their absolute risk, the harder that case becomes.
So it is not simply a question of whether very low apoB is dangerous.
It is whether treating large numbers of healthy people to reach very low apoB is justified by outcomes we have actually shown, rather than outcomes we infer should occur.
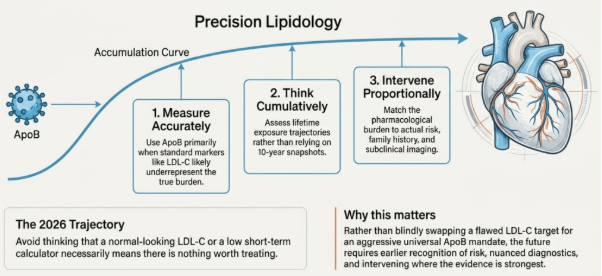
A better model than replacing one threshold with another
The nuance here is not that everyone should be pushed early and hard to a single advanced marker target.
The first step is to measure cardiovascular risk more accurately, think in cumulative exposure rather than only 10-year horizons, use apoB where possible and especially where standard markers are most likely to underrepresent risk, intervene earlier when trajectory, family history, metabolic phenotype, or imaging suggest that the risk is being understated, and keep treatment burden proportional to the quality of evidence and the person in front of the doctor.
Bottom Line
ApoB is closer to the causal biology of atherosclerosis than LDL-C. What follows from that, on current evidence, is earlier recognition of risk more than an aggressive universal target.
No primary-prevention trial has yet shown that treating healthy adults to apoB below 0.6 g/L, for example, produces better outcomes than current practice. Biology, Mendelian randomisation, and discordance data all point in that direction, but we are not there yet with trial data.
The concern is no longer mainly about whether having very low cholesterol is itself harmful. In the populations studied so far, intensive lowering has looked broadly safe, even at very low achieved levels. The real question is a different one: whether the side effects, daily pill burden, cost, and years of taking medication are justified by the actual benefit a person can expect. That calculation depends heavily on underlying risk. The higher someone's lifetime risk of heart disease, the more those trade-offs are worth making. The lower their risk, the harder that case becomes.
So the move is not to replace an LDL-C target with a more aggressive apoB one. It is to measure risk more accurately, think in cumulative exposure rather than 10-year probability, use apoB where standard markers are most likely to understate burden, intervene earlier when trajectory, family history, metabolic phenotype, or imaging warrant it, and keep treatment intensity proportional to both the evidence and the person in the consulting room.
A "normal" LDL-C or a low 10-year risk score is not the same as no risk. But it is also not yet an indication to treat every healthy adult to a single aggressive advanced-marker target.
Q&A
Should I (or my patient) be aiming for apoB below 0.6 g/L? Not as a universal target. The figure is biologically attractive and genetically coherent, but no primary-prevention trial has tested whether treating healthy adults to below 0.6 g/L produces better outcomes than current practice. In people at genuinely high lifetime risk (familial hypercholesterolaemia, established atherosclerosis, long-standing diabetes with other adverse features) aggressive targets are more defensible. In low-risk healthy adults, the target should be set as part of a broader conversation about lifetime risk, not applied by default.
Does this mean younger adults should be started on lipid-lowering therapy earlier? In some circumstances, yes, and the 2026 guideline explicitly moves in that direction for familial hypercholesterolaemia, markedly raised LDL-C, or strong family history. It does not follow that every younger adult with a mildly raised apoB should be on long-term medication. The argument for earlier intervention is strongest when the lifetime exposure trajectory is clearly unfavourable, not when the short-term score is simply low.
Is the 10-year risk calculator obsolete? No. It remains useful for estimating near-term probability, particularly in older adults where the short-term horizon and absolute risk align more closely. Its limits are specific: it gives little weight to cumulative burden in younger people, and it can understate risk in those whose LDL-C looks reassuring but whose particle number does not. Guidelines are increasingly pairing it with lifetime-risk models and, in selected cases, with coronary artery calcium imaging or apoB.
If intensive lowering looks safe in trials, why is long-term treatment burden still a concern? Two reasons. Most trials run for a few years in higher-risk patients, not decades in low-risk ones, so their reassurance does not automatically extend to 30+ years of treatment in a younger, healthier population. And the safety of an achieved low lipid level is a separate question from the side-effect, adherence, cost, and medicalisation burden of the medications that produce it. A reassuring answer to the first question does not settle the second.
How should the decision to treat actually be made? By putting apoB in context rather than treating the number in isolation. Lifetime risk, family history, metabolic phenotype, and imaging where available should carry more weight than any single marker. The greater the lifetime risk, the more the case for earlier and more intensive intervention. The weaker it is, the harder it becomes to justify the long-term treatment burden. ApoB is a better measurement. It is not, on its own, a decision.
Further Reading
[1] A. D. Sniderman et al., ‘Apolipoprotein B Particles and Cardiovascular Disease: A Narrative Review’, JAMA Cardiol., vol. 4, no. 12, pp. 1287–1295, Dec. 2019, doi: 10.1001/jamacardio.2019.3780.
[2] ‘Physiological Bases for the Superiority of Apolipoprotein B Over Low‐Density Lipoprotein Cholesterol and Non–High‐Density Lipoprotein Cholesterol as a Marker of Cardiovascular Risk | Journal of the American Heart Association’. Accessed: Apr. 23, 2026. [Online]. Available: https://www.ahajournals.org/doi/10.1161/JAHA.122.025858
[3] T. G. Richardson et al., ‘Evaluating the relationship between circulating lipoprotein lipids and apolipoproteins with risk of coronary heart disease: A multivariable Mendelian randomisation analysis’, PLoS Med., vol. 17, no. 3, p. e1003062, Mar. 2020, doi: 10.1371/journal.pmed.1003062.
[4] N. A. Marston et al., ‘Association of Apolipoprotein B–Containing Lipoproteins and Risk of Myocardial Infarction in Individuals With and Without Atherosclerosis: Distinguishing Between Particle Concentration, Type, and Content’, JAMA Cardiol., vol. 7, no. 3, pp. 250–256, Mar. 2022, doi: 10.1001/jamacardio.2021.5083.
[5] A. D. Sniderman, L. Dufresne, K. M. Pencina, S. Bilgic, G. Thanassoulis, and M. J. Pencina, ‘Discordance among apoB, non–high-density lipoprotein cholesterol, and triglycerides: implications for cardiovascular prevention’, Eur. Heart J., vol. 45, no. 27, pp. 2410–2418, Jul. 2024, doi: 10.1093/eurheartj/ehae258.
[6] Cholesterol Treatment Trialists’ (CTT) Collaboration et al., ‘Efficacy and safety of more intensive lowering of LDL cholesterol: a meta-analysis of data from 170,000 participants in 26 randomised trials’, Lancet, vol. 376, no. 9753, pp. 1670–1681, Nov. 2010, doi: 10.1016/S0140-6736(10)61350-5.
[7] C. T. T. (CTT) Collaborators, ‘The effects of lowering LDL cholesterol with statin therapy in people at low risk of vascular disease: meta-analysis of individual data from 27 randomised trials’, The Lancet, vol. 380, no. 9841, pp. 581–590, Aug. 2012, doi: 10.1016/S0140-6736(12)60367-5.
[8] Writing Committee Members et al., ‘2026 ACC/AHA/AACVPR/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Dyslipidemia: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines’, Circulation, vol. 0, no. 0, doi: 10.1161/CIR.0000000000001423.
[9] J. Shepherd et al., ‘Pravastatin in elderly individuals at risk of vascular disease (PROSPER): a randomised controlled trial’, The Lancet, vol. 360, no. 9346, pp. 1623–1630, Nov. 2002, doi: 10.1016/S0140-6736(02)11600-X.
[10] S. D. Wiviott et al., ‘Can low-density lipoprotein be too low? The safety and efficacy of achieving very low low-density lipoprotein with intensive statin therapy: a PROVE IT-TIMI 22 substudy’, J. Am. Coll. Cardiol., vol. 46, no. 8, pp. 1411–1416, Oct. 2005, doi: 10.1016/j.jacc.2005.04.064.
[11] R. P. Giugliano et al., ‘Long-term Safety and Efficacy of Achieving Very Low Levels of Low-Density Lipoprotein Cholesterol : A Prespecified Analysis of the IMPROVE-IT Trial’, JAMA Cardiol., vol. 2, no. 5, pp. 547–555, May 2017, doi: 10.1001/jamacardio.2017.0083.
[12] M. L. O’Donoghue et al., ‘Long-Term Evolocumab in Patients With Established Atherosclerotic Cardiovascular Disease’, Circulation, vol. 146, no. 15, pp. 1109–1119, Oct. 2022, doi: 10.1161/CIRCULATIONAHA.122.061620.
[13] R. P. Giugliano et al., ‘Cognitive Function in a Randomized Trial of Evolocumab’, N. Engl. J. Med., vol. 377, no. 7, pp. 633–643, Aug. 2017, doi: 10.1056/NEJMoa1701131.
[14] ‘Atorvastatin Reduces First and Subsequent Vascular Events Across Vascular Territories: The SPARCL Trial’, J. Am. Coll. Cardiol., Accessed: Apr. 20, 2026. [Online]. Available: https://www.jacc.org/doi/10.1016/j.jacc.2020.03.015
[15] P. Amarenco et al., ‘Benefit of Targeting a LDL (Low-Density Lipoprotein) Cholesterol <70 mg/dL During 5 Years After Ischemic Stroke’, Stroke, vol. 51, no. 4, pp. 1231–1239, Apr. 2020, doi: 10.1161/STROKEAHA.119.028718.
[16] D. A. Rudolph et al., ‘Association of Long-term Statin Use With the Risk of Intracerebral Hemorrhage: A Danish Nationwide Case-Control Study’, Neurology, vol. 99, no. 7, pp. e711–e719, Aug. 2022, doi: 10.1212/WNL.0000000000200713.