BPC-157 and the Difference Between an Evidence Gap and a Cover-Up: What the entire human evidence base actually looks like, and the questions to ask next.
In April 2026, the UK medicines regulator opened an investigation into peptide clinics making health claims about compounds they cannot legally sell as medicines [1]. BPC-157 was named first. The MHRA's stated read is that there is little credible scientific evidence behind the claims, the products are unlicensed, and the marketing has run a long way ahead of the data.
If you have spent any time inside the longevity or biohacker side of the internet over the past two years, you have heard the opposite story. BPC-157 heals tendons. Closes ulcers. Repairs the gut. Restores joint function. Speeds recovery. Some clinicians and podcasters speak about it as if it were a quietly acknowledged frontier therapy that regulators are sitting on for reasons of patent, profit, or institutional suppression.
Both cannot be true. This piece is about which one is, and why the answer matters beyond a single peptide. It is not about whether peptide therapeutics as a class are interesting. They absolutely are. It is about whether this peptide, now, justifies the way it is being sold.
What BPC-157 actually is
BPC-157 is a synthetic 15-amino-acid peptide. Its sequence corresponds to a fragment of a larger protein originally identified in human gastric juice [2]. The molecule sold today is not extracted from anyone's stomach. It is manufactured by solid-phase synthesis, typically as a research chemical.
Its preclinical biology is genuinely interesting. In rodent models, BPC-157 accelerates tendon healing, gastric ulcer closure, skin and muscle wound repair, and recovery from several forms of induced injury [2]. Mechanistically, it upregulates VEGFR2 signalling, stimulates angiogenesis and fibroblast proliferation, and modulates the inflammatory cytokine response [3]. It is also unusually stable in gastric acid, which is why oral dosing is even theoretically plausible.
That is the legitimate basis for interest. None of it is fabricated. The problem is what happens next.
What is biologically interesting is not yet clinically justified
In medicine, "biologically interesting" and "clinically justified" are not the same statement. Conflating them is exactly how unlicensed peptides get sold to people who would not otherwise buy them.
Consider the human evidence base for BPC-157 in full.
There is one open-label intravenous safety pilot in two healthy adults, who received single doses of 10 mg and 20 mg with no comparator, no blinding, and short-term observation limited to vital signs, self reported symptoms and labs across two days [4].
There is one retrospective chart review of sixteen patients with chronic knee pain who paid for intra-articular injections. Fourteen reported subjective improvement, but there was no control group, no blinded assessment, no objective functional endpoint and no follow-up imaging endpoint [5].
There is one retrospective chart review of twelve women with severe interstitial cystitis who received a single treatment session of BPC-157 injections into the bladder [6].
There is one Phase I trial registered as NCT02637284 in 2015, intended to enrol forty-two participants, which was subsequently cancelled with no published results [7].
That is the entire human dataset. No placebo control. No blinded assessment. No standardised endpoint. There is no published, peer-reviewed, full-paper randomised human trial with accessible results showing that BPC-157 is effective or safe for any indication. All three published full-paper human studies include Edwin Lee as lead author. None of them addresses the actual claim being sold to biohackers, health conscious individuals or athletes which is that BPC-157 enhances recovery, repair, or longevity in healthy people.
Decades of rodent work do not transfer into a clinical recommendation by default. They tell you a molecule is worth the cost and complexity of a proper human trial. They do not substitute for one.
But a Phase II randomised placebo-controlled hamstring-strain trial (NCT07437547) is registered, with no published results yet [8].
Three questions are worth carrying over to the next unlicensed peptide somebody markets to you. How many randomised, placebo-controlled human trials exist in your specific population? Where does the available human evidence sit on the study design hierarchy: case report (low grade), retrospective series, or controlled trial (higher grade)? Who funded the existing evidence, and do they sell the product? If those answers are all unsatisfactory, the molecule is at the same maturity stage as BPC-157, regardless of how compelling the rodent data look.
The suppression story, taken seriously
The strongest version of the suppression argument is not nonsense, and it should not be dismissed.
It runs roughly as follows. BPC-157's amino acid sequence is in the public domain. There is no composition-of-matter patent worth pursuing. The molecule is small, cheap to synthesise, and would be difficult to defend commercially. Bringing any drug through Phase II and Phase III to regulatory approval costs hundreds of millions. No commercial sponsor will fund that for a compound it cannot exclusively monetise. Therefore the trials will not happen, and BPC-157 will sit in regulatory limbo while clinics sell it under the "research use only" loophole.
That part is structurally accurate. The economics of pharmaceutical development do create real evidence gaps for unpatentable molecules, and patients pay the cost of that asymmetry.
What does not follow is the conspiracy. The cancelled Phase I trial was not buried by a regulator; it was abandoned, almost certainly because no sponsor was willing to underwrite the next phase.
The FDA concern is not just “lack of approval”. It is a manufacturing and safety problem.
Peptides are chains of amino acids, and their safety depends heavily on purity, identity, stability and route of administration. If a compounded product contains peptide fragments, impurities, aggregates, degradation products, or an incompletely characterised active ingredient, the immune system may recognise parts of that product as foreign. That is what their reference to "immunogenicity" means. The clinical consequences could range from local inflammation or rash through to serious health issues, such as systemic hypersensitivity, including anaphylaxis, or antibody formation. Those antibodies could make future doses less effective, increase the risk of future reactions, or, in some therapeutic proteins, interfere with related natural proteins in the body.
Because BPC-157 lacks robust human safety data across the proposed routes of administration, the FDA states it does not have enough information to know whether compounded BPC-157 would cause harm in humans. The basis is evidence absence, not commercial threat [9]. The MHRA's current stance is built on the same gap.
The honest version of the economic argument is: we have an evidence gap because the incentives are broken. It does not require a cover-up. It requires philanthropic, academic, or government-funded trials, which is a tractable problem rather than a sinister one.
The suppression frame matters because it mis-directs attention. A reader who believes evidence is being hidden looks for the smoking gun. A reader who understands the actual structural problem can ask better questions of any unpatentable therapeutic, and tell promising-but-untrialled apart from suppressed-but-proven. The two are not the same.
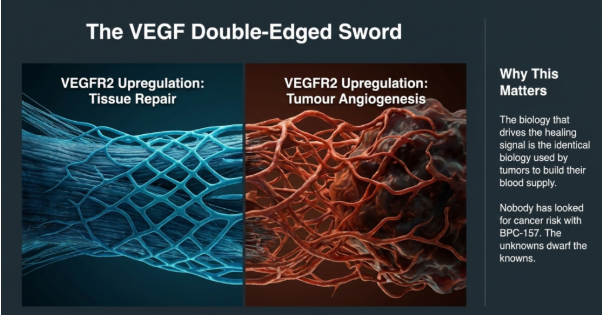
The real, untold risk
One of the reasons BPC-157 has attracted attention is its potential to support new blood vessel growth during tissue repair. In animal studies, it appears to influence pathways linked to VEGF, a key signal involved in healing [10], [11].
The same VEGF pathways are also used by tumours to build their own blood supply [12]. No human data exist on tumour-related risk for BPC-157 in either direction. That is not a claim that BPC-157 causes cancer. It is the more serious statement that nobody has looked, and the biology that drives the healing signal is the same biology that warrants the question. Even if short-term studies report no safety concerns, they would not be designed to detect longer-term risks like cancer or abnormal tissue growth, which may take years to emerge.
Immunogenicity is unmeasured. Repeated administration of any synthetic peptide can provoke antibody formation, and the chronic dosing schedules sold by clinics have not been characterised in humans for this molecule [9]. Product purity is unverified. Grey-market BPC-157 is sold without independent certificates of analysis, and the FDA has specifically flagged contamination, mischaracterisation, and impurity risk for compounded peptides as a class.
For competitive athletes there is a separate, definitive problem: WADA classifies BPC-157 as an S0 prohibited substance, meaning any in-competition or out-of-competition use is sanctionable [13]. For UK clinics selling it as a treatment, the MHRA position now creates direct legal exposure [1].
The shape of all this is not "BPC-157 is dangerous." It is that the unknowns dwarf the knowns, and that asymmetry is the position. Nobody can give you an honest probability of harm because the studies that would generate one have not been done.
Where the credible stance sits, and what would change it
Someone looking to explore peptides must ensure they have some reliable and independent way of understanding the evidence base and the risks in doing so.
There is a narrow case where use of BPC-157 might be defensible. A patient with a serious non-healing musculoskeletal injury, conventional options exhausted, full informed consent, ideally inside a registered investigational protocol with structured safety monitoring. That is a rare clinical conversation. It is not the use case being marketed to members of the public looking for faster recovery from training, or to biohackers looking for a longevity edge. Those are different conversations.
What would move this position is: a randomised, placebo-controlled trial in a defined population with an objective primary endpoint, a pre-registered protocol, and pharmacovigilance over a clinically meaningful duration. Phase II at minimum, with a credible safety analysis. None of that exists today. If it does in five years, the position would change.
The discipline of waiting for trials, rather than substituting rodent data and case-series enthusiasm for them, is responsible and credible clinical judgement. Holding that discipline is what ensures first do no harm remains central, particularly in health-conscious individuals where potential benefit is uncertain and avoidable risk matters more. The MHRA, in April 2026, has effectively made the same call.
Q&A
Q: BPC-157 is a fragment of a protein found naturally in the stomach. Doesn't that make it inherently safe?
This is the most common misconception. The naturally occurring parent protein sits in the stomach environment in low concentrations and acts locally in the stomach. The product sold by clinics is a synthetic 15-amino-acid peptide manufactured by "solid-phase synthesis", administered under the skin, into the muscle or a joint, or intravenously, often at higher doses for weeks. A "natural" origin of a substance tells you nothing about overall safety at a synthetic dose, by an unphysiological route, in a manufactured form whose purity is not independently verified.
Q: Does the evidence support using BPC-157 for tendon or musculoskeletal injuries?
Not yet, and the gap is specific. Rodent models show accelerated tendon healing, increased collagen organisation, and improved biomechanical strength under controlled injury conditions. The single human dataset for musculoskeletal use is a retrospective chart review of sixteen knee-pain patients who paid for intra-articular injections, with subjective improvement reported in fourteen and no control, no blinded assessment, and no objective imaging endpoint. A Phase II placebo-controlled hamstring-strain trial (NCT07437547) is now registered, with no published results. What the evidence supports today is biological plausibility worth a proper trial; it does not support a clinical recommendation, including for athletes with tendon injuries.
Q: If BPC-157 is genuinely promising, why hasn't a proper trial been done?
The reason is unapologetically unglamorous and worth understanding. The amino acid sequence is in the public domain and there is no composition-of-matter patent worth pursuing, so no commercial sponsor will fund the hundreds of millions a Phase II–III development programme costs for a compound it cannot exclusively monetise. The cancelled 2015 Phase I trial (NCT02637284) was almost certainly abandoned for this reason rather than buried by a regulator. Trials of unpatentable molecules typically depend on philanthropic, academic, or government funding, and that pipeline has not yet materialised for BPC-157.
Q: What about buying research-grade BPC-157 from a reputable supplier? Doesn't that solve the quality problem?
It does not, and the FDA's reasoning here is worth taking seriously. "Research use only" labelling is a legal classification, not a quality guarantee, and there is no mandated independent assay of identity, purity, aggregation state, or contamination for products in this market. The FDA's concerns cite peptide manufacturing risks: incompletely characterised active ingredient, peptide fragments, degradation products, and impurities that the immune system may recognise as foreign. The clinical consequence ranges from local inflammation through to systemic hypersensitivity and antibody formation. A clean Certificate of Analysis from the supplier is not the same as an independent third-party assay, and almost no buyer is in a position to audit the difference.
Q: Is there any clinical scenario in which BPC-157 use is defensible?
A narrow one. A patient with a serious non-healing musculoskeletal injury, conventional options exhausted, full informed consent, and ideally enrolment in a registered investigational protocol with structured safety monitoring. That is a rare clinical scenario and it is not the use case being marketed to the public. For recovery enhancement, performance optimisation, or longevity goals in healthy people, until a randomised, placebo-controlled trial in a defined population with an objective primary endpoint and pharmacovigilance over a clinically meaningful duration produces data we do not currently have, we just don't have good evidence of benefit, let alone safety.
Further Reading
[1] N. Davis and S. Marsh, ‘Medicines watchdog to investigate UK peptide clinics over health claims’, The Guardian, Apr. 04, 2026. Accessed: Apr. 29, 2026. [Online]. Available: https://www.theguardian.com/society/2026/apr/04/medicines-watchdog-to-investigate-uk-peptide-clinics-over-health-claims
[2] PubChem, ‘Bpc-157’. Accessed: Apr. 29, 2026. [Online]. Available: https://pubchem.ncbi.nlm.nih.gov/compound/9941957
[3] F. P. McGuire, R. Martinez, A. Lenz, L. Skinner, and D. M. Cushman, ‘Regeneration or Risk? A Narrative Review of BPC-157 for Musculoskeletal Healing’, Curr. Rev. Musculoskelet. Med., vol. 18, no. 12, pp. 611–619, Dec. 2025, doi: 10.1007/s12178-025-09990-7.
[4] E. Lee and K. Burgess, ‘Safety of Intravenous Infusion of BPC157 in Humans: A Pilot Study’, Altern. Ther. Health Med., vol. 31, no. 5, pp. 20–24, Sep. 2025.
[5] E. Lee and B. Padgett, ‘Intra-Articular Injection of BPC 157 for Multiple Types of Knee Pain’, Altern. Ther. Health Med., vol. 27, no. 4, pp. 8–13, Jul. 2021.
[6] E. Lee, C. Walker, and B. Ayadi, ‘Effect of BPC-157 on Symptoms in Patients with Interstitial Cystitis: A Pilot Study’, Altern. Ther. Health Med., vol. 30, no. 10, pp. 12–17, Oct. 2024.
[7] PharmaCotherapia d.o.o., ‘Phase I, Pilot Study in Healthy Volunteers, to Assess the Safety and Pharmacokinetics of PCO-02, Which Active Ingredient is BPC-157, a Penta-deca-peptide From Gastric Source.’, clinicaltrials.gov, Clinical trial registration NCT02637284, Dec. 2015. Accessed: Apr. 29, 2026. [Online]. Available: https://clinicaltrials.gov/study/NCT02637284
[8] Hudson Biotech, ‘A Randomized, Double-Blind, Placebo-Controlled Phase 2 Trial of Pentadecapeptide BPC 157 for Accelerated Repair of Acute Grade II Hamstring Strain Confirmed by MRI’, clinicaltrials.gov, Clinical trial registration NCT07437547, Feb. 2026. Accessed: Apr. 29, 2026. [Online]. Available: https://clinicaltrials.gov/study/NCT07437547
[9] C. for D. E. and Research, ‘Certain Bulk Drug Substances for Use in Compounding that May Present Significant Safety Risks’, FDA, Apr. 2026, Accessed: Apr. 29, 2026. [Online]. Available: https://www.fda.gov/drugs/human-drug-compounding/certain-bulk-drug-substances-use-compounding-may-present-significant-safety-risks
[10] L. Brcic, I. Brcic, M. Staresinic, T. Novinscak, P. Sikiric, and S. Seiwerth, ‘Modulatory effect of gastric pentadecapeptide BPC 157 on angiogenesis in muscle and tendon healing’, J. Physiol. Pharmacol. Off. J. Pol. Physiol. Soc., vol. 60 Suppl 7, pp. 191–196, Dec. 2009.
[11] M.-J. Hsieh et al., ‘Therapeutic potential of pro-angiogenic BPC157 is associated with VEGFR2 activation and up-regulation’, J. Mol. Med., vol. 95, no. 3, pp. 323–333, Mar. 2017, doi: 10.1007/s00109-016-1488-y.
[12] D. Hanahan and R. A. Weinberg, ‘Hallmarks of cancer: the next generation’, Cell, vol. 144, no. 5, pp. 646–674, Mar. 2011, doi: 10.1016/j.cell.2011.02.013.
[13]‘BPC-157: Experimental Peptide Creates Risk for Athletes’. Accessed: Apr. 29, 2026. [Online]. Available: https://www.usada.org/spirit-of-sport/bpc-157-peptide-prohibited/